
[ad_1]
Credit: Sales reports (2024). DOI: 10.1016/j.celrep.2024.113740
Toxoplasmosis is a worldwide infectious disease caused by the single-celled parasite Toxoplasma gondii. In humans, infection poses a particular risk to pregnant women, as it can cause birth defects. Like the closely related malaria pathogen — Plasmodium falciparum — and other related species, T. gondii has specialized organelles, so-called rhoptries and micronemes, to infect the host cell.
A team led by Professor Marcus Meissner, Chair of Experimental Parasitology at LMU, and Professor Joel Dix of the University of Alberta (Canada) has now investigated the evolution of this infection machinery and identified an organelle-specific protein, a Could be a promising target. for new treatment methods. Paper Published in Sales reports.
During their evolution, parasites developed not only specialized organelles such as rhoptries and micronemes, but all Protein machinery Essential to ensure the production and function of organelles.
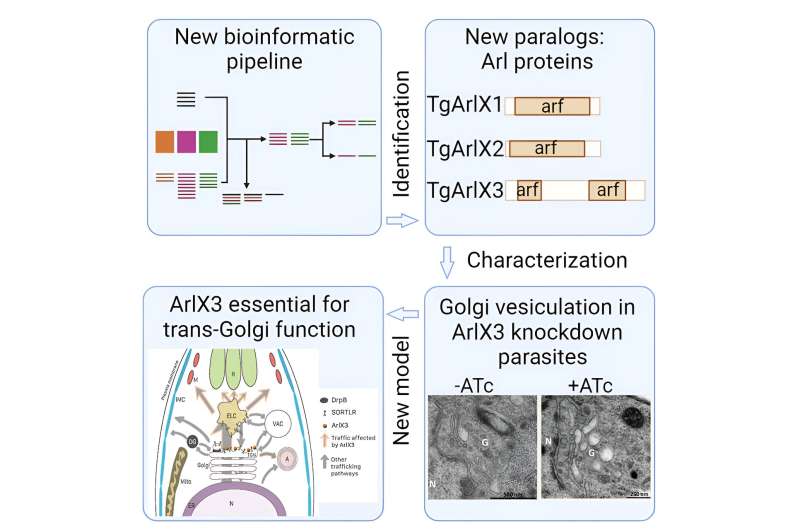
The so-called organelle paralogy hypothesis (OPH) proposes that the current diversity of cell organelles is due to the duplication and subsequent diversification during evolutionary time of certain genes encoding organelle identity.
“To build their specific structures, the parasites had to reproduce some proteins and add others,” explains LMU parasitologist Elena Jimenez-Ruiz.
A combination of bioinformatics and lab analyses
To investigate how and to what extent organelle diversity evolved in the Apicomplexa—the group of organisms to which T. gondii and P. falciparum belong—the researchers used comprehensive bioinformatic genetic analyzes and molecular cell biology methods. combined with In this way, the scientists were able to identify 18 candidate proteins involved in the emergence of new organelles and their protein machinery apicomplexan.
“For one of these proteins, known as ArlX3, we were able to show experimentally that it plays a key role in the formation of micronemes and rhoptries in T. gondii,” Jimenez-Ruiz said. say “Without ArlX3, parasites cannot reproduce.”
Because ArlX3 occurs in parasites but not in human cells, the researchers believe the protein may represent a promising target structure for new therapeutic options. Moreover, their methods could potentially be used to identify more specific protein subgroups that could yield important insights into parasite cell biology.
More information:
Christine M. Klinger et al., Evolutionary analysis identifies the Golgi pathway and links lineage-specific factors to endomembrane organelle emergence in the apicomplexans, Sales reports (2024). DOI: 10.1016/j.celrep.2024.113740
Provided by
Ludwig Maximilian University of Munich
Reference: Toxoplasmosis: Researchers identify protein that evolved with infection machinery (2024, Feb 16) Accessed 18 Feb 2024 at https://phys.org/news/2024-02-toxoplasmosis-protein-evolved-infection-machinery. Retrieved from html
This document is subject to copyright. No part may be reproduced without written permission, except for any fair dealing for the purpose of private study or research. The content is provided for informational purposes only.
[ad_2]


