
[ad_1]
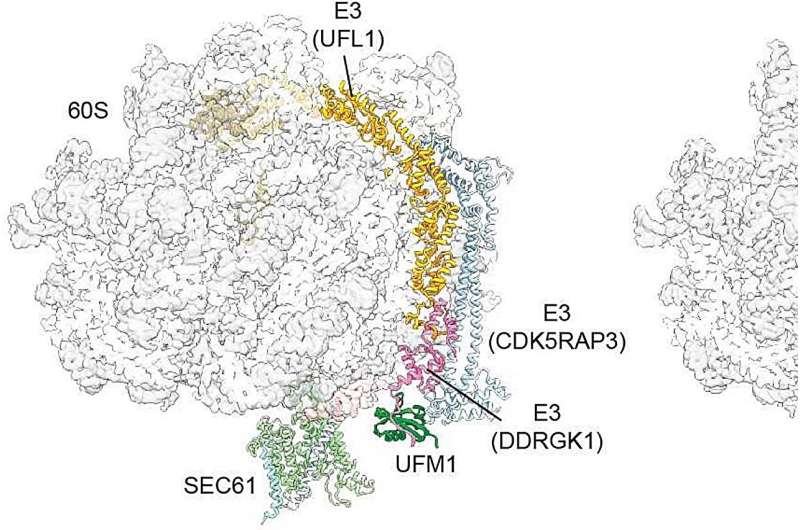
The protein UFM1 acts as a molecular wedge, facilitating cleavage of the Sec channel. Credit: T. Baker, Nature 2024
Protein synthesis in the cell is an important process of life. Through this, the genetic code of the genome is translated into the amino acid sequence of proteins. This process is complex and has been studied in detail for decades.
Protein biosynthesis is carried out by specialized molecular machines, The ribosome, which consists of a large and a small subunit. At the end of Protein biosynthesisthese protein factories have to be broken down into their individual parts (recycled), so that they are ready for the next cycle. translation.
Now a team led by Prof. Roland Beckmann, Dr. Thomas Becker, and Ivan Panchio from LMU’s Gene Center Munich, working with researchers at Stanford University led by Prof. Ron Kopito, has shown How the so-called ribosome recycling Endoplasmic reticulum (ER) functions.
In the process, they discovered the role of an enzyme, a special E3 ligase that links a small protein modification called UFM1 to the large ribosomal subunit, as a key recycling mechanism. An account of their investigation is given. published I The nature.
Detailed insights into ribosome recycling
Ribosomes are normally found floating within the cytoplasm. “Here we know exactly how recycling works,” says Baker. Sometimes, however, they are located on a continuous cell-wide membrane network in the ER.
Although many proteins are produced in the cytosol, they must later be transported to other organelles, such as mitochondria, chloroplasts, and many others. If a protein is synthesized in the ER membrane, the entire translation machinery is submerged in the ER membrane. This is accomplished with the help of a protein-conducting channel (SEC61), which is able to transport proteins across the membrane or insert them into the membrane during synthesis.
There is another one after the completion of the translation. recycling step, which is specific to ER. membrane. That is, the large subunit of the ribosome has to be dissociated from the protein-conducting channel again.
Beckman’s team has now shown how this subunit works: When translation is terminated, the E3 ligase recognizes the large subunit of the ribosome. “It—figuratively speaking—places a small wedge, the protein UFM1, in the large subunit,” Becker explains.
“This creates a stable complex between the modified 60S subunit and the E3 ligase. At the same time, it causes the large subunit to dissociate from SEC61. This is a very important step in ensuring that the large subunit The unit is returned to the cytosol and is available for the next cycle.”
More information:
Paul A. DaRosa et al, UFM1 E3 ligase promotes recycling of 60S ribosomal subunits from the ER, The nature (2024). DOI: 10.1038/s41586-024-07073-0
Provided by
Ludwig Maximilian University of Munich
Reference: Researchers Reveal How Cells Regenerate Protein Factories in Endoplasmic Reticulum (2024, February 23)
This document is subject to copyright. No part may be reproduced without written permission, except for any fair dealing for the purpose of private study or research. The content is provided for informational purposes only.
[ad_2]


